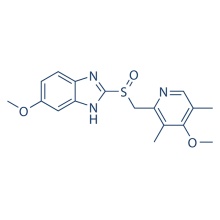
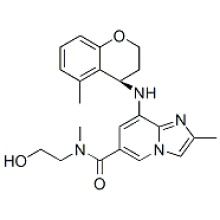
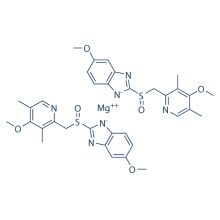
Click on the follow link to find out more information: https://www.medicine-voice.com/respiratory-digestive-system-drug/
Company Info
- Company Name: NINGBO VOICE BIOCHEMIC CO. LTD
- Representative: Quan Sheng Ren
- Product/Service: Oncology Anticarcinogenic , Antipyretic Analgesics , Antiparasitic & Antimycotic , Digestive System Drug , Neural System Remedy , Respiratory System Remedy
- Capital (Million US $): 5000000RMB
- Year Established: 2015
- Export Percentage: 61% - 70%
- Contact Person: Mr. Medic Ren
- Tel: 86-574-87330957
Other Products
Hot Products
Astragaloside AChlortetracycline HCl 64-72-2Paclitaxel 33069-62-4Dexamethasone Acetate 1177-87-3Dinaciclib (SCH727965) 779353-01-4CHIR-124 405168-58-3Ro3280 1062243-51-9TAME 901-47-3CCG-1423 285986-88-110058-F4 403811-55-2Dabigatran (BIBR 953) 211914-51-1H 89 2HCl 130964-39-5T0901317 293754-55-9Aprepitant 170729-80-3Turofexorate Isopropyl (XL335) 629664-81-9BMS-378806 357263-13-9