Thiazovivin 1226056-71-8
Product Description
.cp_wz table {border-top: 1px solid #ccc;border-left:1px solid #ccc; } .cp_wz table td{border-right: 1px solid #ccc; border-bottom: 1px solid #ccc; padding: 5px 0px 0px 5px;} .cp_wz table th {border-right: 1px solid #ccc;border-bottom: 1px solid #ccc; padding: 5px 0px 0px 5px;}
Molecular Weight:
311.36 Thiazovivin is a novel ROCK Inhibitor with IC50 of 0.5 μM, promotes hESC survival after single-cell dissociation.
Biological Activity
Although displaying little impact on cell proliferation, Thiazovivin
treatment significantly enhances the survival of human embryonic stem
cells (hESCs) after enzymatic dissociation more than 30-fold, while
homogenously maintaining pluripotency with the characteristic colony
morphology, expression of typical pluripotency markers such as alkaline
phosphatase (ALP), and normal karyotype. Dissociated hESCs treated with
Thiazovivin display dramatically increased adhesion to matrigel- or
laminin-coated plates but not to gelatin-coated plates within a few
hours. Thiazovivin treatment increases cell-ECM adhesion-mediated β1
integrin activity, which synergizes with growth factors to promote cell
survival.
In addition to activating integrin, Thiazovivin but not
Tyrintegin (Ptn) protects hESCs from death in the absence of ECM in
suspension through E-cadherin-mediated cell-cell interaction.
Thiazovivin treatment potently inhibits endocytosis of E-cadherin,
consequently stabilizing E-cadherin on the cell surface and leading to
reestablishment of cell-cell interaction, which is essential for hESC
survival in ECM-free conditions.
Thiazovivin but not Tyrintegin (Ptn) at
2 μM inhibits Rho-associated kinase (ROCK) activity and protects hESCs
at a similar level as the widely used selective ROCK inhibitor Y-27632
at 10 μM, suggesting that Rho-ROCK signaling regulates cell-ECM and
cell-cell adhesion. Thiazovivin at 1 μM increases the reprogramming
efficiency of CB mononuclear cells to induced pluripotent stem cells
(iPSCs) by more than 10 times.
Protocol(Only for Reference)
Kinase Assay:
[1]
Conversion of different model animals based on BSA (Value based on data from FDA Draft Guidelines)
For example, to modify the dose of resveratrol used for a mouse (22.4 mg/kg) to a dose based on the BSA for a rat, multiply 22.4 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for resveratrol of 11.2 mg/kg.
Chemical Information
Molarity Calculator
Dilution Calculator
Molecular Weight Calculator
Contact us if you need more details on 1226056-71-8. We are ready to answer your questions on packaging, logistics, certification or any other aspects about Thiazovivin 1226056-71-8、1226056-71-8 Thiazovivin. If these products fail to match your need, please contact us and we would like to provide relevant information.
Molecular Weight:
311.36 Thiazovivin is a novel ROCK Inhibitor with IC50 of 0.5 μM, promotes hESC survival after single-cell dissociation.
Biological Activity
Although displaying little impact on cell proliferation, Thiazovivin
treatment significantly enhances the survival of human embryonic stem
cells (hESCs) after enzymatic dissociation more than 30-fold, while
homogenously maintaining pluripotency with the characteristic colony
morphology, expression of typical pluripotency markers such as alkaline
phosphatase (ALP), and normal karyotype. Dissociated hESCs treated with
Thiazovivin display dramatically increased adhesion to matrigel- or
laminin-coated plates but not to gelatin-coated plates within a few
hours. Thiazovivin treatment increases cell-ECM adhesion-mediated β1
integrin activity, which synergizes with growth factors to promote cell
survival.
In addition to activating integrin, Thiazovivin but not
Tyrintegin (Ptn) protects hESCs from death in the absence of ECM in
suspension through E-cadherin-mediated cell-cell interaction.
Thiazovivin treatment potently inhibits endocytosis of E-cadherin,
consequently stabilizing E-cadherin on the cell surface and leading to
reestablishment of cell-cell interaction, which is essential for hESC
survival in ECM-free conditions.
Thiazovivin but not Tyrintegin (Ptn) at
2 μM inhibits Rho-associated kinase (ROCK) activity and protects hESCs
at a similar level as the widely used selective ROCK inhibitor Y-27632
at 10 μM, suggesting that Rho-ROCK signaling regulates cell-ECM and
cell-cell adhesion. Thiazovivin at 1 μM increases the reprogramming
efficiency of CB mononuclear cells to induced pluripotent stem cells
(iPSCs) by more than 10 times.
Protocol(Only for Reference)
Kinase Assay:
[1]
| In vitro ROCK assay | Thiazovivin is dissolved in DMSO. CycLex Rho-kinase assay kit is used to detect ROCK activity using recombinant ROCK in the presence of increasing concentrations of Thiazovivin (~10 μM). |
|---|
Conversion of different model animals based on BSA (Value based on data from FDA Draft Guidelines)
| Species | Baboon | Dog | Monkey | Rabbit | Guinea pig | Rat | Hamster | Mouse |
| Weight (kg) | 12 | 10 | 3 | 1.8 | 0.4 | 0.15 | 0.08 | 0.02 |
| Body Surface Area (m2) | 0.6 | 0.5 | 0.24 | 0.15 | 0.05 | 0.025 | 0.02 | 0.007 |
| Km factor | 20 | 20 | 12 | 12 | 8 | 6 | 5 | 3 |
| Animal A (mg/kg) = Animal B (mg/kg) multiplied by | Animal B Km |
| Animal A Km |
For example, to modify the dose of resveratrol used for a mouse (22.4 mg/kg) to a dose based on the BSA for a rat, multiply 22.4 mg/kg by the Km factor for a mouse and then divide by the Km factor for a rat. This calculation results in a rat equivalent dose for resveratrol of 11.2 mg/kg.
| Rat dose (mg/kg) = mouse dose (22.4 mg/kg) × | mouse Km(3) | = 11.2 mg/kg |
| rat Km(6) |
Chemical Information
| Molecular Weight (MW) | 311.36 |
|---|---|
| Formula | C15H13N5OS |
| CAS No. | 1226056-71-8 |
| Storage | 3 years -20℃Powder |
|---|---|
| 6 months-80℃in solvent (DMSO, water, etc.) | |
| Synonyms | |
| Solubility (25°C) * | In vitro | DMSO | 15 mg/mL (48.17 mM) |
|---|---|---|---|
| Water | <1 mg/mL ( | ||
| Ethanol | <1 mg/mL ( | ||
| In vivo | 30% PEG400/0.5% Tween80/5% propylene glycol | 30 mg/mL | |
| * <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. | |||
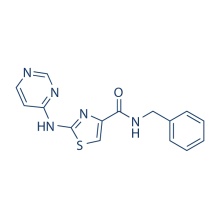
| Chemical Name | N-benzyl-2-(pyrimidin-4-ylamino)thiazole-4-carboxamide |
|---|
Molarity Calculator
Dilution Calculator
Molecular Weight Calculator
Contact us if you need more details on 1226056-71-8. We are ready to answer your questions on packaging, logistics, certification or any other aspects about Thiazovivin 1226056-71-8、1226056-71-8 Thiazovivin. If these products fail to match your need, please contact us and we would like to provide relevant information.
Product Categories : Cell Cycle > ROCK Inhibitor
Other Products
Hot Products
Astragaloside AChlortetracycline HCl 64-72-2Paclitaxel 33069-62-4Dexamethasone Acetate 1177-87-3Dinaciclib (SCH727965) 779353-01-4CHIR-124 405168-58-3Ro3280 1062243-51-9TAME 901-47-3CCG-1423 285986-88-110058-F4 403811-55-2Dabigatran (BIBR 953) 211914-51-1H 89 2HCl 130964-39-5T0901317 293754-55-9Aprepitant 170729-80-3Turofexorate Isopropyl (XL335) 629664-81-9BMS-378806 357263-13-9